Explanation About Osmosis
Explanation About Osmosis, Indeed recently has been hunted by consumers around us, perhaps one of you personally. People now are accustomed to using the internet in gadgets to view video and image information for inspiration, and according to the name of this article I will discuss about
If the posting of this site is beneficial to our suport by spreading article posts of this site to social media marketing accounts which you have such as for example Facebook, Instagram and others or can also bookmark this blog page.
The process important in biology was first thoroughly studied in 1877 by a german plant physiologist wilhelm pfeffer.
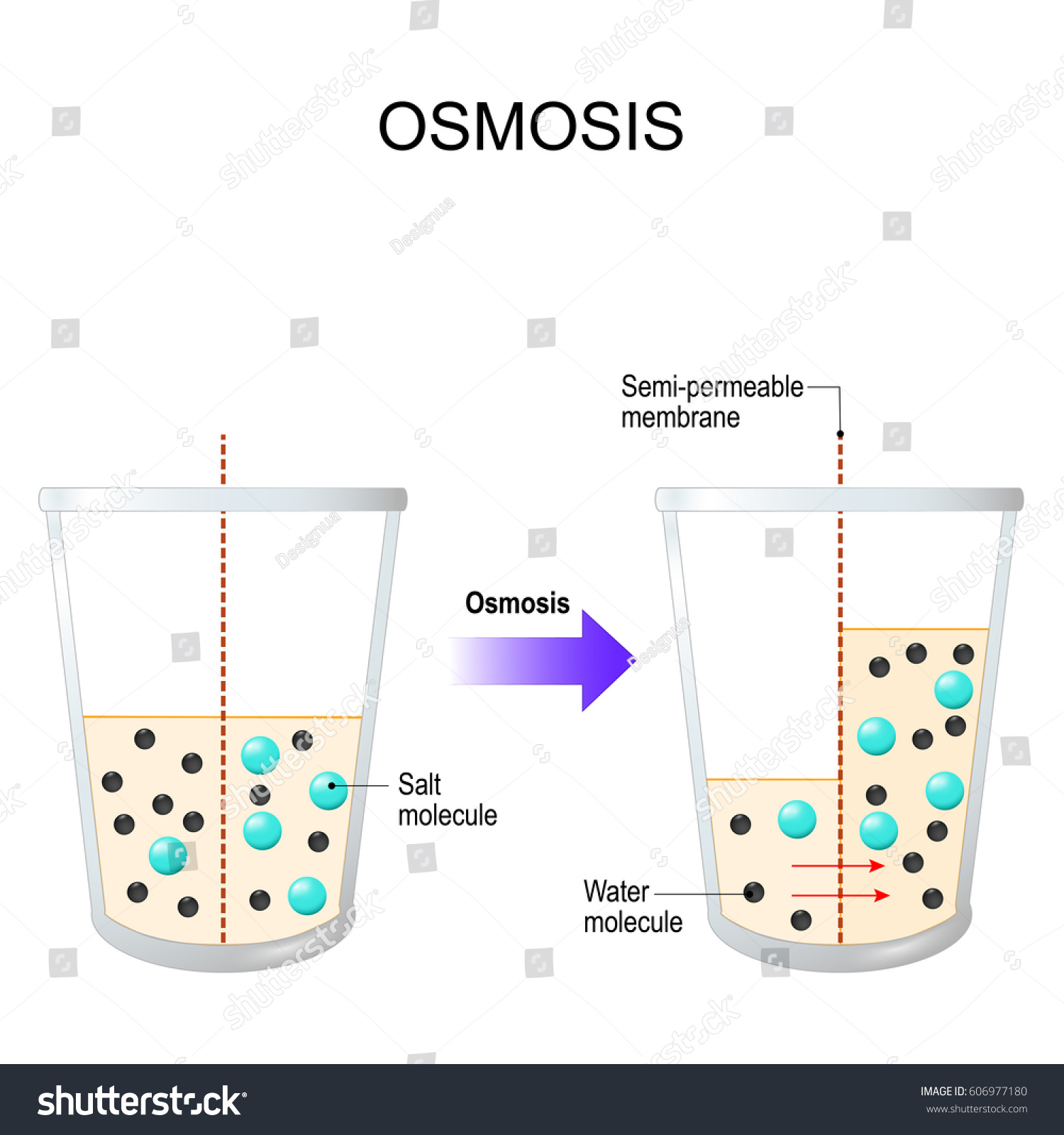
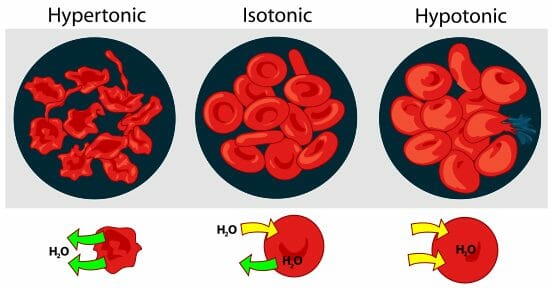
Arti kata explanation text. Hypertonic isotonic and hypotonic solutions and their effect on cells. If youre seeing this message it means were having trouble loading external resources on our website. Osmosis can be defined as the movement of water from where it is in higher concentration to where it is in lower concentration through a semi permeable membrane.
Osmosis definition is movement of a solvent such as water through a semipermeable membrane as of a living cell into a solution of higher solute concentration that tends to equalize the concentrations of solute on the two sides of the membrane. To make it easier for you imagine there is a cup which has water in it. It should swell up a little this is because the water.
Osmosis is the movement of water from a less concentrated solution to a more concentrated. Osmosis is in short the process by which water diffuses through a membrane. Some kinds of osmotic flow have been observed since ancient times.
However there are several factors that contribute to the direction of which osmosis occurs. When water moves passively through a cell membrane thats osmosis. By definition osmosis is the movement of any solvent through a selectively permeable membrane into an area of higher solute concentration the result of which will be an equalizing of solute concentration on either side of the membrane.
Osmosis is when a substance crosses a semipermeable membrane in order to balance the concentrations of another substance. Try soaking a raisin in water what happens. Osmosis happens spontaneously and without any energy on the part of the cell.
Osmosis the spontaneous passage or diffusion of water or other solvents through a semipermeable membrane one that blocks the passage of dissolved substancesie solutes. As mentioned before osmosis may be. In biology this is usually when a solvent such as water flows into or out of a cell depending on the concentration of a solute such as salt.
The main constraint on osmosis is concentration. A semi permeable membrane is any membrane that allows only certain substances to pass through it. Osmosis refers to the movement of fluid across a membrane in response to differing concentrations of solutes on the two sides of the membrane.
The endosmometer invented by dutrochet. The removal of water from a tissue by salt was referred to as imbibition. Osmosis has been used since antiquity to preserve foods by dehydration with salt or sugar.